Abstract
Hydrogen energy has been proposed as a reliable and sustainable source of energy which could play an integral part in demand for foreseeable environmentally friendly energy. Biomass, fossil fuels, waste products, and clean energy sources like solar and wind power can all be employed for producing hydrogen. This comprehensive review paper provides a thorough overview of various hydrogen storage technologies available today along with the benefits and drawbacks of each technology in context with storage capacity, efficiency, safety, and cost. Since safety concerns are among the major barriers to the broad application of H2 as a fuel source, special attention has been paid to the safety implications of various H2 storage techniques. In addition, this paper highlights the key challenges and opportunities facing the development and commercialization of hydrogen storage technologies, including the need for improved materials, enhanced system integration, increased awareness, and acceptance. Finally, recommendations for future research and development, with a particular focus on advancing these technologies towards commercial viability.
An overview of hydrogen storage technologies – Key challenges and opportunities
Keywords
Hydrogen ,Hydrogen storage,Metal hydride,SWOT analysis,Sustainable aspects,Future prospects,Challenges Opportunities
1. Introduction
1.1. Background
The industrial revolution has been pushing the world's energy consumption by manifolds. Our reliance on the dwindling fossil fuels will pose a challenge to meet the exponential growth in energy requirements. Additionally, these fossil fuels pose a great threat to the environment by increasing CO2 in the atmosphere, which is the leading cause of air pollution and deterioration of human health. These have necessitated us to look for renewable energy sources to meet the demands. Among many renewable energy sources, hydrogen energy has been recognized as a potential replacement for fossil fuels . It is accepted as a promising energy vector because of its ability to provide superior energy service efficiently and sustainably . Furthermore, hydrogen can be produced by a variety of non-fossil and fossil resources, making it highly versatile.
A researcher at the International Institute for System Analysis in Austria named Marchetti argued for H2 economy in an article titled “Why hydrogen” in 1979 based on proceeding 100 years of energy usage . The essay made predictions, which have been referenced in studies on the H2 economy, that have remarkably held concerning the consumption of coal, natural gas, as well as oil during the subsequent 40 years . Although the authors did not foresee a drop in nuclear investment or a delay in the development of fusion technology, the emergence of natural gas as the world's main source of energy has mostly followed their predictions. The case for a hydrogen economy put out by Marchetti and others today is strengthened by the decline in nuclear technology and the requirement to replace fossil fuels .
Furthermore, this move towards a hydrogen economy is met with many technological challenges. The current production method employed for hydrogen is natural gas reforming, which is not environmentally friendly. The non-fossil fuel method for hydrogen production mainly using solar energy is still in the development phase and is critical for the hydrogen economy.
The most effective way to make this change is through using hydrogen as an alternative fuel because it is an "ideal energy carrier" and is both sustainable as well as environmentally friendly. Massive H2 production and effective H2 delivery and storage remain the main obstacles to its use, nevertheless .depicts hydrogen and its routes.
1.2. Properties and applications of hydrogen
The smallest and most plentiful is hydrogen, which possesses a proton, an electron, and no neutrons in its most common form (1H, protium). Hydrogen is also the most lightweight element. Nuclear fusion devices use the other two naturally occurring isotopes 2H (deuterium, 3), which has one proton and one neutron within the nucleus, and 3H (tritium), which has one proton and two neutrons .
The element that is most prevalent in the universe is hydrogen. Hydrogen is thought to make up around 91 % of the cosmos. H2 has been expected by the scientific community as a viable fossil fuel substitute. Clean energy is provided by H2. Fuel cells, which are environmentally beneficial and create H2O as a by-product, are what power H2–powered cars .
One of the main methods for being carbon neutral is to develop hydrogen power. Currently, the lack of safe, effective, as well as affordable hydrogen storage makes industrial-scale hydrogen.
application impractical. Hydrogen is frequently liquefied or compacted to improve its density since it has a low volumetric energy density (0.0899 kg/m3) under atmospheric circumstances. However, these technologies have enormous prices, and safety concerns, and call for large storage systems. They don't meet the requirements for hydrogen storage as a result. Therefore, materials–based hydrogen storage has garnered a lot of interest [
15].
In comparison to other sources of energy currently available, both chemical and physical qualities have made it extremely difficult to store and transport affordably for wide application in recent years [
16]. Due to H2's low volumetric and energy densities at standard temperature, compacted pressure of 700 bar, and liquid states (0.0107 MJ/L and 5.6 MJ/L, respectively) as well as its higher heating value of 143 MJ/kg, the storage method is incredibly challenging .
Among all fuels, H2 was found to be the most energetic element per kg of 120 MJ/kg (Abe et al., 2019), and when burned, the only exhaust by-product is water .
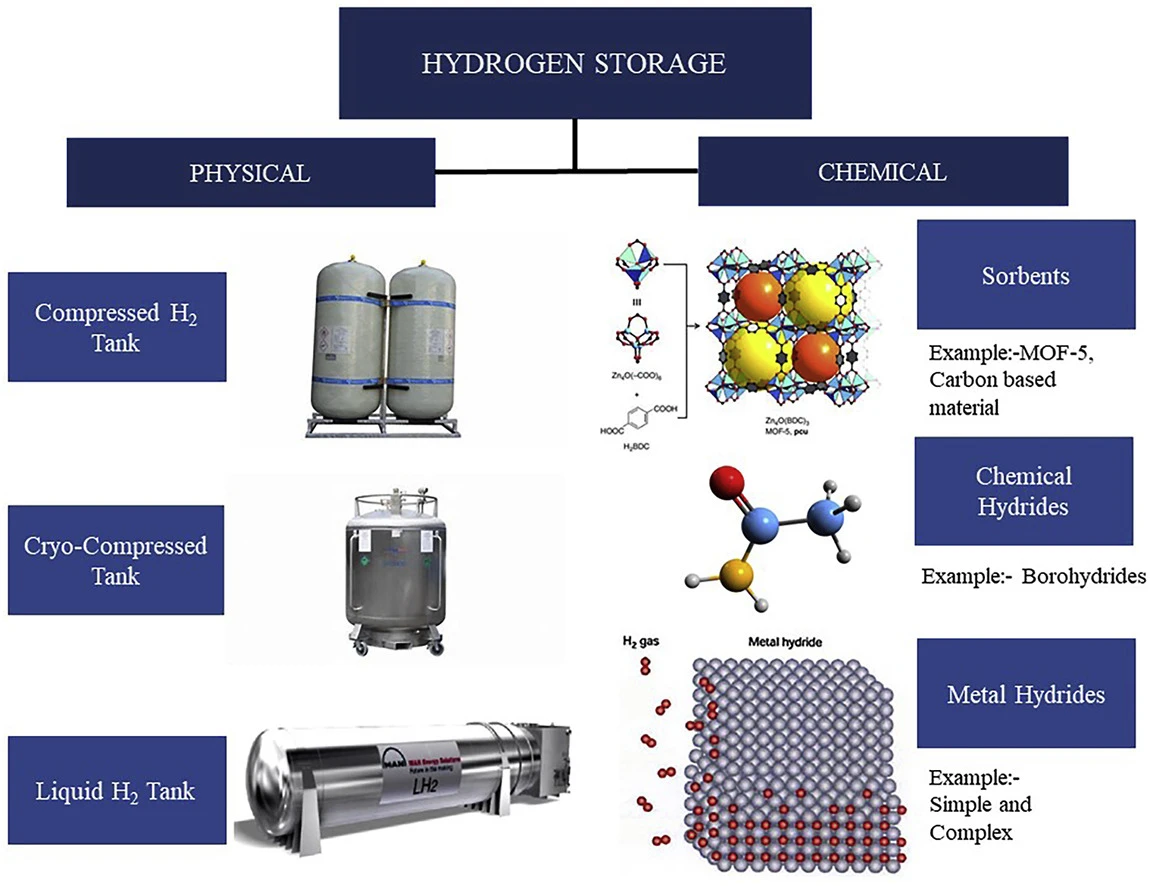
It is difficult to store H2 since it is a gas at ambient temperature and under air pressure. As a result of its low density (0.089 kg/m3 under normal circumstances). Despite having a higher gravimetric energy density than fossil fuels due to being the lightest element, H2 gas has a far lower volumetric energy density. Different H2 storage systems, including high–pressure gas storage, low – temperature liquid storage, solid-state storage, and liquid organic storage, have been developed to address this issue .
With a liquid density of 70.8 kg/m3 at standard boiling point pressure of 1 atm and a vapor density of 0.08376 kg/m3 at 20 °C and 1 atm . H2 is lighter than other gases present in air and it gets easily dispersed in air. Due to its variety of synthesis methods and sources of energy (such as light and heavy hydrocarbon oils, solar, wind, geothermal, nuclear, biofuels as well as biogas), it is frequently referred to as an efficient energy carrier rather than a power source .